 
Due to this reason elements on the right side of the periodic table are small in size because they experienced the higher effective nuclear charge.ī) Trend of the periodic table in the group:- The atomic radii of elements increases from top to bottom in a group. Thus, effective nuclear charge (Zeff) increases across the period from left to right. As a result, electrons are pulled closer to the nucleus by the increased effective nuclear charge and thereby, cause a decrease in the size of the atom. Since the electrons in the same shell do not screen each other from the nucleus, the increase in nuclear charge is not neutralized by the extra valence electron. 
With the increase in atomic number from lithium to fluorine, the magnitude of the nuclear charge increases progressively by one unit while the corresponding addition of electrons takes place in the same principal shell. This is explained based on increasing nuclear charge along a period. For example, in the second period, the atomic radii decrease from Li to Ne through Be, C, N, O, and F as given in the table This means that the electrons in the inner orbital s screen the electrons in the valence shell more effectively than p orbital and so on.Ī) Trend of the periodic table in period:- In general, the atomic radii decrease with increase (from left to right) in a period. Order of screening constant of orbital is: Shielding Constant / Screening constant: – It is the repulsive force experienced by the valence shell from the inner electrons. 
Nuclear charge (Z):- It is the total number of protons present in the nucleus. For example, the metallic radius of sodium is 186 pm whereas the covalent radius of sodium is just 154 pm.Įffective Nuclear Charge (Zeff):-It is the charge experienced by an electron in a polyelectronic atom after the screening effect of the inner shell electron. (iii) Metallic Radius: It is the minimum distance between two nuclei of metal ions when they are adjacent to each other in a metal lattice. So, the Van der Waals radius of the chlorine is 180 pm. For example, the internuclear distance of two chlorine atoms is 360 pm. 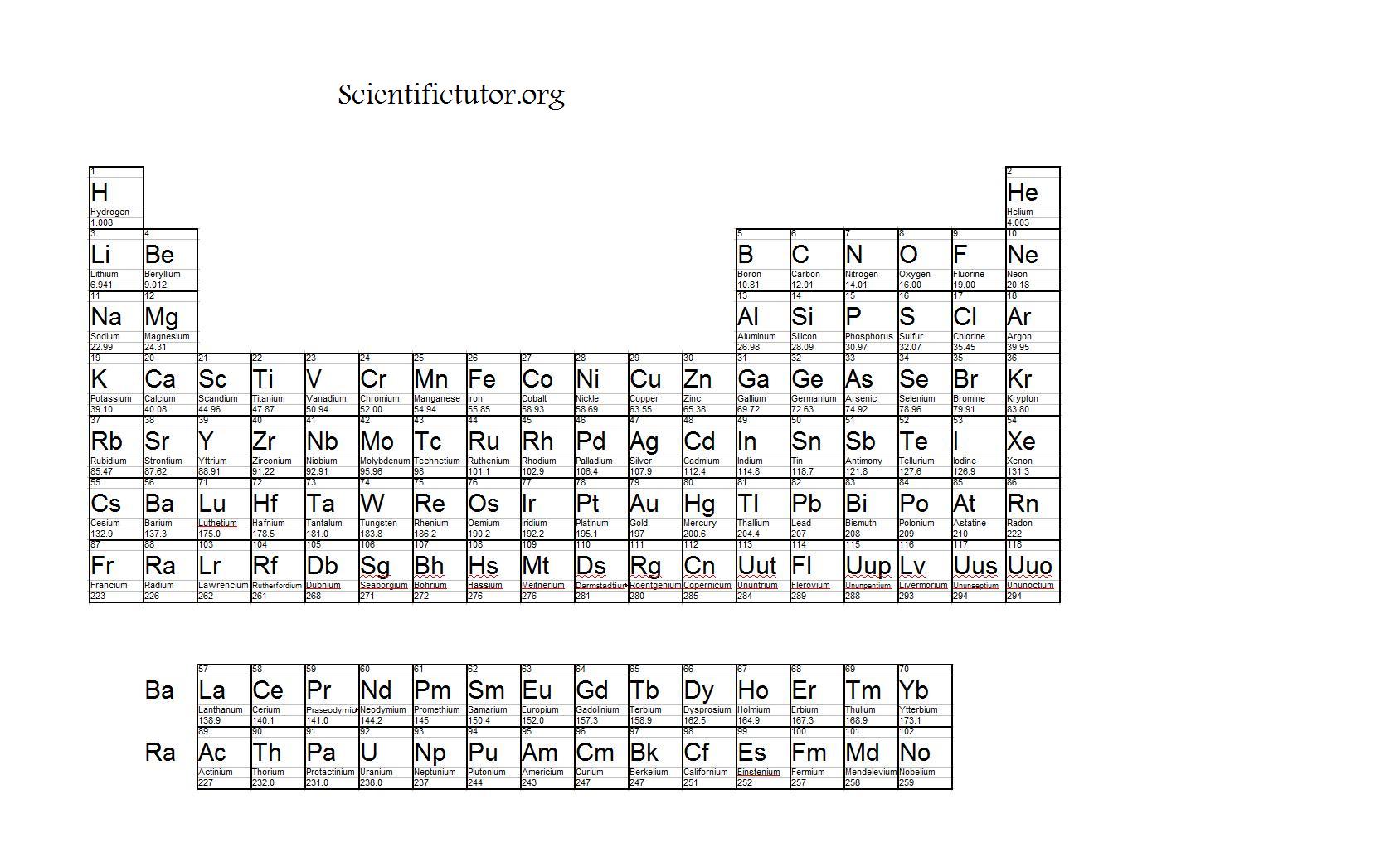
(ii) Van der Waals radius: It is half of the distance between the two atoms belonging to the neighboring molecules. For covalently bonded atoms we use a covalent radius. (i) Covalent radius: It is half of the distance between two covalently joined atoms. In this article, we will learn more about Atomic radii, Types of atomic radii, Trends of atomic radii, Effective nuclear charge and Shielding constant. Thus electrons are added in that outer orbital which increases the distance from the nucleus. Because of two reasons, one of them is that the atomic number increases down the group so, the positive nuclear charge increases, And other is, due to an increase in principal quantum number (n).Ītomic radius is the distance between the nucleus and the outermost shell of the electron.Īs we move in the periodic table from one element to the next across a period, nuclear charge (Z) increases by one, but screening due to inner electrons increases only slightly.ĭown the group, atomic radii increase because of the addition of an extra shell. From top to bottom in a group, the size of atoms generally increases. But there are some exceptions observed such as the atomic radius of the oxygen atom is slightly more than the radius of the nitrogen atom. From left to right, the period size of the atoms generally decreases. As we move in the periodic table from top to bottom in groups and left to right in periods, a change in the atomic radius is observed. When two similar atoms are joined together by the bond then the atomic radius is one-half of the distance between the nuclei of the two atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
